by Emma Kampouraki
While a vast proportion of our knowledge about drugs focuses on the mechanism of action, the indications and the adverse events that may appear, we are still at an early stage to understand fully the interaction between drugs and our genetic background. It is well established that such interaction is present and has a great impact on the response of patients to a number of commonly used prescription medication.
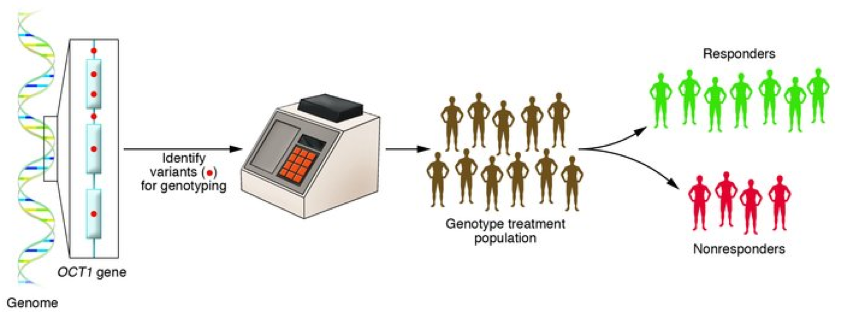
After the observation of substantial differences between patients’ responses, while receiving the same treatment, it became apparent that the “one size fits all” design is not effective anymore. Vogel in 1959, described the term ‘pharmacogenetics’ as the field that studies the way in which one single change in a gene influences the response to a drug. However, after the completion of the Human Genome project in 2003, the term ‘pharmacogenomics’ was used to describe the influence of the whole genome to the drug response. Pharmacogenetics is a subspecialty of Pharmacogenomics and does not require whole genome sequencing, whereby analysis of all the genes is performed. The two terms are often used interchangeably, but this sometimes can cause confusion.
The importance of pharmacogenetics is also reflected in the recent clinical trials. There is an increasing interest to study how drug responses are affected by patients’ genetic variations as demonstrated by the 1060 studies about pharmacogenetics or pharmacogenomics to date.
There are numerous examples of drugs that are influenced by certain genes, either regarding their pharmacodynamics or pharmacokinetics. One important gene that encodes a microsomal liver enzyme from the P450 complex is CYP2D6. As shown in 1970, this gene affects the elimination of debrisoquine, an antihypertensive drug. People carrying certain alleles (i.e. versions) of this gene have altered elimination rates and therefore different duration of action.
Also, certain alleles of the NAT2 gene lead to the production of less N-acetyltransferase which causes slower acetylation and elimination in patients and side effects from the longer stay of the drug in the blood circulation.
Similarly, suxamethonium, a general anaesthetic, can cause prolonged apnoea in patients with defects in BCHE gene producing low activity of the enzyme that normally breaks down suxamethonium. Primaquine, a drug against malaria, has been shown to cause acute haemolysis in patients with G6PD enzyme deficiency.
The most important example is that of warfarin. This anticoagulant drug has a very narrow therapeutic window, causing serious, potentially lethal side effects after small instabilities in its levels. The dose for each patient has been proven to be dependent on two genes, CYP2C9 that affects the elimination by metabolising the S- enantiomer of warfarin and VKORC1, the actual target enzyme that warfarin inhibits.
In fact, FDA updated the drug labelling in 2007, recommending very specific dose adjustments after genetic testing of these two genes in combination.
In order to organise the information from the studies, a database was created, called PharmaGKB, which can easily facilitate the acquisition of information among researchers and clinicians and the actual clinical implementation of pharmacogenetics.
So, what exactly are the benefits from translating pharmacogenetic information into clinical practice? In a study of 2008, it was estimated that over one fourth of commonly prescribed drugs have some type of genetic information that could be used in medicine. It is possible to predict interactions between drugs, potential side-effects and individualization of treatment, such as informed choice of drugs or dosage as in the case of warfarin.
Currently, several kits for pharmacogenetic testing for warfarin are commercially available with their cost continuously decreasing, making genetic testing easier than ever before. In Freeman Hospital, Newcastle upon Tyne, such a kit utilises literally a droplet of patient blood and provides the genetic analysis result within 40 minutes after a fully automated run.
A more personalized design of new drugs, especially in population level, can be achieved using the different allele frequencies in genes that are important for drug activity and metabolism. This way, each population would receive more appropriate drugs schemes for its most prevalent genetic changes. Although there is a great number of challenges in this field, including ethics and concern about potential access to the data, which can be partially addressed by the advances of technology, the benefits are many and the cost-effectiveness of the implementation of pharmacogenetics is reported in many studies. The field of pharmacogenetics has therefore a lot to offer to the scientific and medical community.