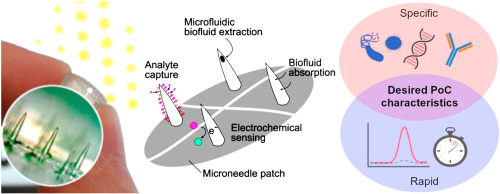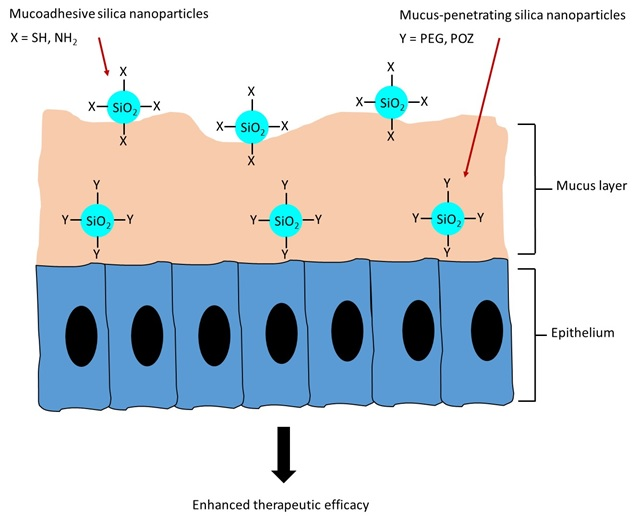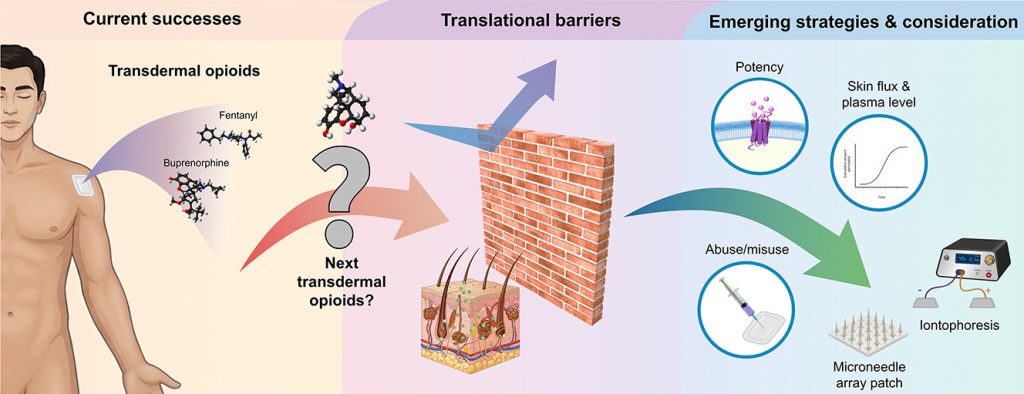
We have a new review article published in the Journal of Controlled Release: https://doi.org/10.1016/j.jconrel.2026.114894
Liang YM, Azizi J, Ng KW, Goh CF. Transdermal delivery and formulation design of opioids: Where are we now and where are we heading? J Control Release. 2026;394:114894. https://doi.org/10.1016/j.jconrel.2026.114894
This has been another excellent transnational collaboration with Dr Choon Fu Goh and colleagues. In this paper, we take a deep dive into a seemingly simple question with a deceptively complex answer: why is it so difficult to deliver opioid drugs across the skin?
Transdermal delivery offers clear advantages for opioid therapy, including more stable drug levels and improved patient convenience compared to oral or injectable routes. However, only a small number of opioids, such as fentanyl and buprenorphine, have been successfully developed into marketable patches. The main limitation is the skin itself. As a barrier, it is highly effective, and drugs require a very specific balance of properties, including molecular size, lipophilicity, and potency, to cross it. Many widely used opioids, including morphine, fall outside this narrow window.
In the review, we examine the main strategies that have been explored to address this challenge, including chemical permeation enhancers, formulation approaches, and physical methods. A consistent theme is that success in controlled laboratory settings does not always translate into clinical use. Improving permeation alone is not sufficient if the system cannot deliver clinically relevant doses reliably in practice. It increasingly appears that formulation strategies relying on passive diffusion across intact skin are unlikely to succeed. Looking ahead, active skin penetration strategies are likely to lead progress by enabling more consistent drug delivery while reducing dosing burden.
I thank my co-authors for their contributions to this work, and I hope the review will serve as a useful resource for the research community.