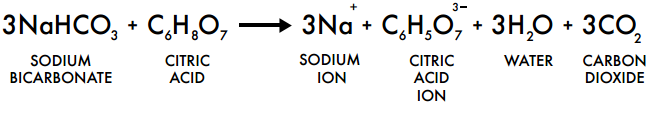Happy Valentine’s Day! Love can confuse your brain, and so does this week’s Try This Tuesday.
You don’t need any equipment to try this experiment at home – you just need to stare at your screen, or more specifically the + in the middle of the picture below. You can blink but don’t look away.

If you stare long enough the pink dots should disappear!
The Science
It looks like the pink dots have disappeared due to a visual phenomenon called Troxler’s fading or Troxler’s effect. if you fix your eyes on a certain point, then anything in your peripheral vision will fade away and disappear after about 20 seconds. In this experiment our sight was focused on the + in the middle of the screen and the the pink dots in your periphery slowly fade and finally disappear. It works especially well in this experiment at there is such low contrast between the light pink dots and the grey background.
This is a type of optical illusion. If you want to see another, have a look at our spinning disk Try This Tuesday.

 1. Add 4 teaspoons of bicarbonate of soda to the glass
1. Add 4 teaspoons of bicarbonate of soda to the glass