Our friends over at the Faculty of Medical Sciences hold an annual series of interactive lectures and practical sessions over six weeks, open to anyone aged 15+ known as Mini Medical School. The aim of the programme is to offer the public an opportunity to find out about current research developments at the university and to learn more about clinical approaches and practices. We asked Heworth Grange student Kate Gordon, 16 to give us her account of the sessions:
Our friends over at the Faculty of Medical Sciences hold an annual series of interactive lectures and practical sessions over six weeks, open to anyone aged 15+ known as Mini Medical School. The aim of the programme is to offer the public an opportunity to find out about current research developments at the university and to learn more about clinical approaches and practices. We asked Heworth Grange student Kate Gordon, 16 to give us her account of the sessions:
I recently attended Newcastle University’s Mini Medical School of 2017. I was overjoyed when I received the confirmation email to say I had a place, however, I didn’t realise just how interesting and beneficial it would be.
“One of the best experiences I’ve had”
When I first arrived I was met by a member of the university team. They were so lovely and pleasant which made my friend and me feel comfortable and welcomed there in their facilities. Not to mention, the great variation of snacks that they provided each week!
On the first week we had Forensic Psychologist, Dr Gavin Oxburgh, speak to us about his part in helping the police with their enquiries. This was the best week for me, as I am very interested in Psychology and it offered insights into a new career path for me later in life. Gavin taught us about offender profiles and how to detect if a person is being deceitful. He also made us think of the circumstances of why people would lie. I really loved this session and even though it was quite a serious topic he made it light-hearted and engaging throughout.

On the second week of my visit Professor Louise Robinson taught us about dementia. This was very interesting for me as quite a lot of my family members suffer from Alzheimer’s disease. We learnt about the 3 different stages and how to prevent memory disorders.
On week 3 we learnt about prescribing drugs and how it’s revolutionised over the years. Dr Adam Todd presented this amazingly and made the topic very humorous. He taught us about opioids and how dangerous they can be. I really loved this session.
During week 4 of mini medical school we were presented with a session about kidney transplantation. As I had already learnt about this topic in biology it really helped as I was able to understand more. We discovered how dialysis has changed throughout the past 50 years and the newer alternatives. This helped me learn a lot more than I already knew.
On the fifth week the topic was named “Sex, Drugs & Rock ‘n’ Roll” this was a good name for the session as it was unknown to what we we’re going to talk about until we arrived. In this session we spoke about HIV and the treatment possible, along with all the other topics we looked at how it’s changed throughout the years. I really liked this evening as Dr Christopher Duncan and Dr Ewan Hunter included humour as well as knowledge.
“There was a range of subjects for everyone which was brilliant as it enabled anyone to participate”
I then attended a mini medical school practical, I chose the topic of Psychology. My day was split into two, first we looked at how the brain worked and the certain areas of it. In the second part we looked at eating disorders and the psychology behind it, for me that was so interesting as it really opened my eyes how to recognise it early, not in only myself but my peers too.
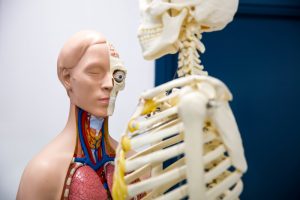
On the sixth and final week we talked about sun protection and dermatological skin problems. The speakers were amazing and worked so well as a team, they told us that even the most famous celebrities suffer from skin diseases such as acne, psoriasis, etc. We also completed a quiz on how to detect cancerous moles, a very helpful technique to use in life. I found this session very interesting and a great way to end the course.
Overall, the Mini Medical School is one of the best experiences I’ve had. I have loved every single minute of it and learnt so much from attending the sessions, it was very enjoyable and didn’t seem like a chore each week – I’m looking forward to next year!
To be added to the mailing list for next years Mini-Medical School, please email minimedicalschool@ncl.ac.uk





 Our friends over at the Faculty of Medical Sciences hold an annual series of interactive lectures and practical sessions over six weeks, open to anyone aged 15+ known as
Our friends over at the Faculty of Medical Sciences hold an annual series of interactive lectures and practical sessions over six weeks, open to anyone aged 15+ known as